Overview
- Glioblastoma, also known as glioblastoma multiforme (GBM) is an aggressive type of cancer that can occur in the brain or spinal cord.
- GBM forms from cells called astrocytes that support other nerve cells.
- Due to the brain’s unique chemical and anatomical features, highly heterogeneous tumor microenvironment, and restricted delivery of chemotherapy or immunotherapy through the blood–brain barrier (BBB), GBM results in a very poor prognosis.
- Conventional methods often fail to predict therapeutic efficacy in GBM, therefore, through 3D bioprinting technologies we can potentially mimic essential physiological and pathological features of GBM and BBB.
- In vitro 3D models of GBM and BBB leveraging patient- or healthy-individual-derived cells and biomaterials through 3D bioprinting technologies, can potentially revolutionise therapies for GBM.
NOTE: ADVANCED BIOENGINEERING MATERIAL IS MARKED ACCORDINGLY. FOR INFORMATION REGARDING THE APPLICATION OF 3D BIO-PRINTING IN DEVELOPING ANTI-CANCER THERAPIES READ THE CONCLUSION SECTION AND WATCH THE VIDEOS IN THE ADDITIONAL RESOURCES SECTION. IF YOU ARE NEW TO THE SUBJECT, WE RECOMMEND READING THE SECTIONS TITLED | GLIOBLASTOMA | AND | CONCLUSION |
| GLIOBLASTOMA |
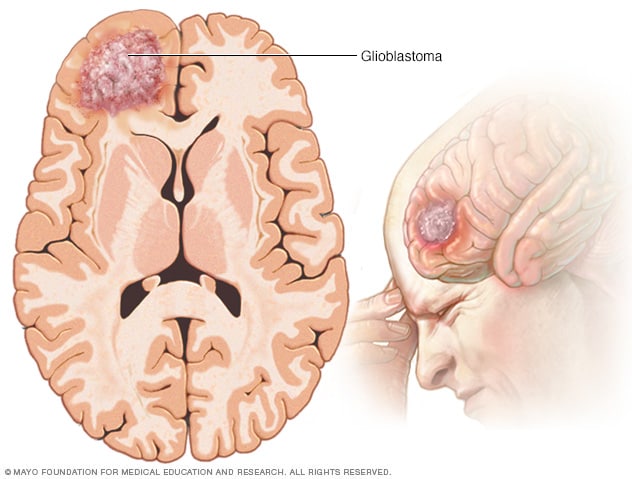
Glioblastoma (GBM) is the most common and aggressive primary brain cancer in adults, accounting for 14.6% of all malignant tumors of the central nervous system (CNS). GBM is a type of astrocytoma. Astrocytomas start in cells called astrocytes. Astrocytes help support nerve cells and transport nutrients to them.
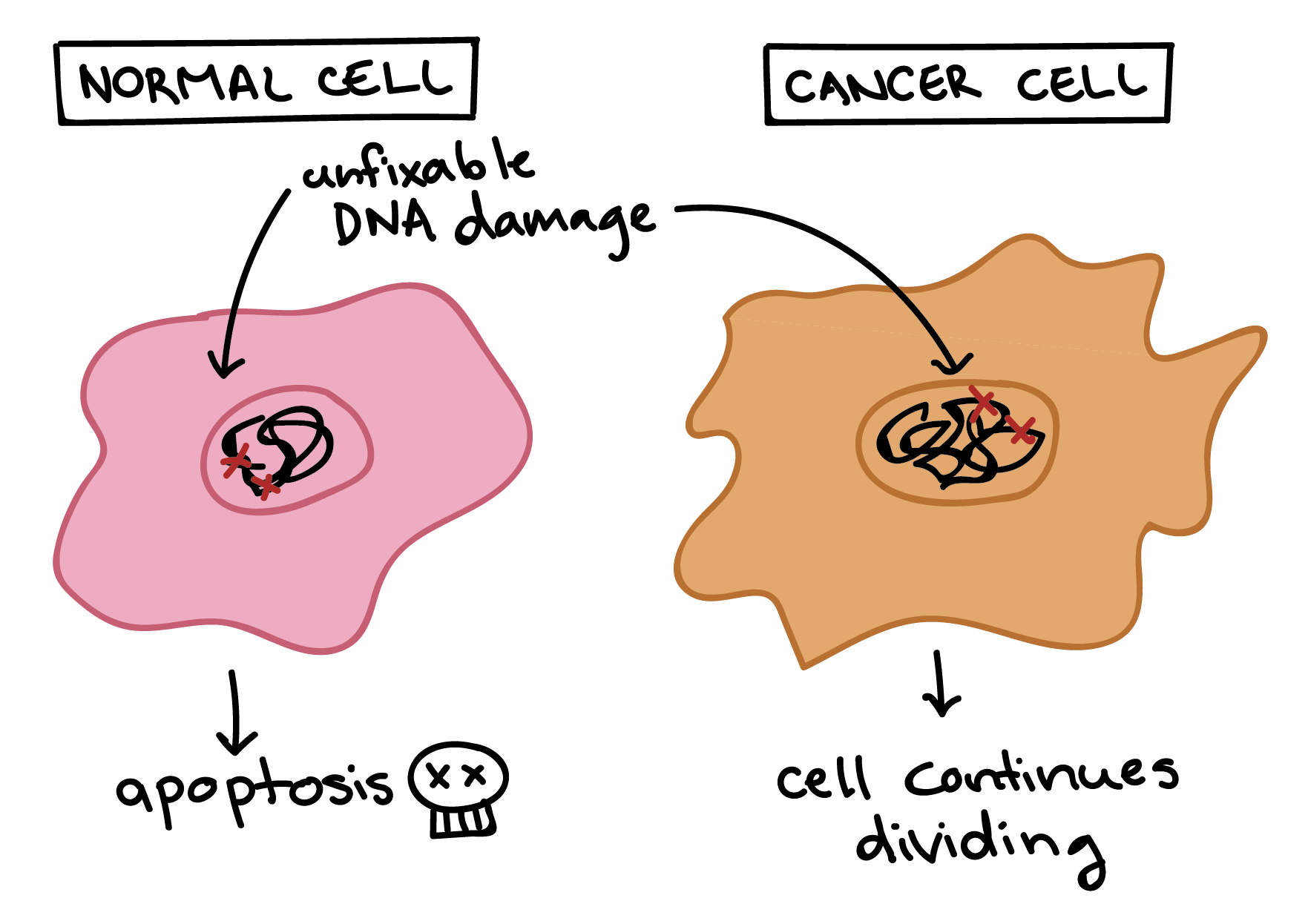
Astrocytes are generally very tightly controlled and organized cells. Something happens that causes them to start multiplying and forming tumors. In simple terms, normal body cells grow and divide and know how to stop dividing when they reach a checkpoint. Overtime, when they have unfixable damage, they die. However, due to some acquired gene mutations, these checkpoints are not understood by cells, and mutations are not noticed. In this case, cells will continue growing and dividing uncontrollably, and become cancer cells. They also do not undergo cell death when they need to.
Some common symptoms of GBM include:
- Headaches
- Loss of appetite
- Loss of balance or trouble walking
- Mood swings
- Nausea and vomiting
- Personality and behavior changes
- Problems speaking
- Problems with memory
- Seizures
- Sensation changes
- Trouble concentrating
- Vision changes
- Weakness
Since initial symptoms such as headaches, nausea and mood swings are generally common ailments among people, GBM can be harder to detect in early stages of tumour formation, which can lead to it growing to a fatal size over time. The 5-year relative survival rate of American patients is 6.8%, ranking the lowest among all primary CNS malignancies.
Despite tremendous efforts in the past decades, little advance in the outcome for patients afflicted with GBM has been achieved. Current treatments include maximal safe surgical removal of the priamry tumour followed by chemoradaition. These methods are usually invasive to the patient and can cause several side effects such as nausea, alopecia, loss of appetite, inflammation and immuno suppression, which can cause adverse results in the patient followed by relapse.
The delivery of therapeutic drugs to GBM tumor sites is particularly challenging compared with other solid tumors because of the limited drug and cell transport across the brain’s only vascular barrier, the blood-brain barrier (BBB). BBB eliminates over 98% of small molecule drugs, and tightly regulates lymphocyte extravasation – movement of immune cells out of the circulatory system and towards the site of tissue damage or infection i.e the tumor. This has several effects on the GBM tissue, including limiting the accumulation of therapeutic agents from chemotherapy, and limiting the presence of active T cells (type of lymphocytes in the immune system that focus on clearing foreign particles). Regulation of BBB or overcoming permeability barriers, facilitates certain therapies against brain tumors, suggesting that the presence of functional BBB may be essential for accurate evaluation of GBM treatments. Side note, Aidan’s PhD research focuses on tmporarily altering BBB permeability for better access of therapeutic agents, using tDCS. The growing interest in replacing current FDA-approved anticancer drugs with improved penetration of BBB for GBM treatments also suggests a potential role for BBB in treatment efficacy.
ADVANCED MATERIAL – GBM MODELLING
In order to make a good GBM model, it is important to encapsulate all the native components of the brain and surrounding tumour microenvironment, the extracellular matrix (ECM) and the BBB which can be summarised using the two images below.


ADVANCED MATERIAL – PRINTING STRATEGIES AND MATERIALS
3D bioprinting is an additive manufacturing technology capable of creating user-defined 3D objects based on computer-aided design (CAD) models. CAD models can be reconstructed from clinical images, such as magnetic resonance imaging (MRI) or computed tomography (CT) scans, or designed using CAD software to represent the specific geometries for individual applications. The 3D models are cut into a series of 2D cross-sections with a predefined layer thickness to be made using a bioprinter. The 3D bioprinting process produces well-defined structures in both three dimensions and high resolution; the reproducibility, flexibility and customizability make it a powerful tool for a wide range of biological applications. To successfully model biological samples, these strategies must allow good cell viability and allow tissues to develop post-printing functions.
Monoculture GBM models enable mechanistic studies and investigations of cell responses to the ECM and the dimensionality. Coculture models allow the investigation of specific cellular interactions between tumor cells and certain stromal components and are good tools for evaluating therapies that regulate these stromal components or their related interactions. Multicellular GBM models capture the highest level of heterogeneity and biomimicry among the in vitro 3D-bioprinted models, thus possessing greater potential as drug screening platforms or for the interrogation of cellular dependencies that have enhanced clinical relevance.
The bioassay of bio-imprinted models requires the use of appropriate biomaterials and combinations of cell types and other relevant molecules (summarised in table 1). The main methods of bioprinting include inkjet bioprinting, extrusion and light-assisted bioprinting processes.

| Material | Type | Crosslinking mechanisms | Common modifications | Composite with other biomaterials | Brain-relevant elastic modulus |
| HA | Natural polysaccharide | Photocrosslinking, shear thinning | Methacrylic anhydride, glycidyl methacrylate, thiol, RGD peptide | Collagen, gelatin, GelMA, chitosan, laminin, fibrin, PEG, PU | 11 Pa to 3.5 kPa |
| Gelatin | Natural protein | Thermal, photocrosslinking, enzymatic | Methacrylate | HA, PU, collagen, PEGDA, fibrin, alginate, chitosan, fibrinogen | 0.49–12.8 kPa |
| dECM | Natural mixture | Relies on composite material | – | Collagen | 78.09 ± 29.22 Pa |
| Collagen | Natural protein | Thermal, Photocrosslinking | Methacrylate | HA, GelMA, fibrin, agarose, riboflavin | 0.9–3.6 kPa |
| Matrigel | Natural mixture | Thermal | – | PEG, gelatin, alginate, agarose | 0.4 kPa |
| Fibrin | Natural protein | Enzymatic | – | HA, collagen, laminin | 0.058–4 kPa |
| Silk fibroin | Natural protein | Photocrosslinking, thermal | Methacrylate | Collagen, gelatin | 17.1 ± 7.8 kPa |
| Gellan gum | Natural protein | Calcium ions, photocrosslinking | RGD peptide, methacrylate | GelMA | 6.4–17.2 kPa |
| PNIPAAm | Synthetic polymer | Thermal | – | PEG | 1.4–3.8 kPa |
| PU | Synthetic polymer | Thermal, Photocrosslinking | – | HA, gelatin | 0.6–8.1 kPa |
| PEG | Synthetic polymer | Photocrosslinking, click chemistry | Methacrylate, thiol, diacrylate, RGD peptide | HA, GelMA, PNIPAAm, laminin | 1–26 kPa |
| SAP | Synthetic peptide | Self-assembly | – | – | 0.3–5.3 kPa |
ADVANCED MATERIAL – PRINTED BBB MODELS
A functional 3D BBB model should summarize the important properties of BBBs, comparable to their physiological levels. The basic characteristics of this vascular barrier include tightness, integrity, selective permeability, and transport mechanisms.
A biological microfluidic device fabricated by two-photon lithography was inoculated with mouse brain endothelial cells and GBM cells. Endothelial cells of the biocombination system form tight junctions and exhibit barrier properties, as verified by ZO1 expression and dextran diffusion (Marino et al, 2018).
Another BBB device assembled with three layers of 3D-printed chambers and a cell insert with iPSC-derived BMECs and astrocytes cultured on both sides of a porous membrane to mimic the architecture initial BBB. A TEER peak of 4000 Ω cm was measured on day 3 of culture, which isoneof the highest Transepithelial/transendothelial electrical resistance (TEER) values reported in in vitro models and in the range of in vivo values. Osmolarities tested with dextrans of different molecular weights and drug compounds correlated with clinical data (Wang et al, 2017).
| CONCLUSION – 3D BIOPRINTING AS A METHOD FOR TRIALING BRAIN ANTI-CANCER THERAPIES |
Failure to treat GBM is the result of multiple factors, including the complex nature of the tumor’s surrounding microenvironment, the rapid progression and treatment resistance of GBM, and the inadequate supply of therapeutic agents to GBM sites due to the barrier nature of the BBB. Trialing new anti-cancer drugs is a very complicated and tightly regulated process, and often takes years of rigorous testing and analyses to develop new therapies. Conducting trials with living beings in itself is a complicated endeavour. Some of the constraints of current research on the BBB can be mitigated using 3D bioprinting strategies. Functional BBBs can be printed in 3D using biomolecules, such as collagen or gelatin. This means, that researchers can use collagen to print a Blood-Brain Barrier that is identical to the one found surrounding our brains. This enables researchers to test the permeability of the BBB while developing new anti-cancer drugs. In other words, they can use these specimens to see if certain drugs will be able to cross the barrier and enter the brain in order to reach the tumor.
The fact that these bioprinted BBBs have not been widely incorporated into clinical research, limits our ability to see how molecules travel from the blood, through the barrier, and into the brain. This also has impacts on treatment outcomes, because the current methods in use take a very long time to be properly tested.
Advances in 3D bioprinting and artificial biomaterials provide clinically relevant modeling capabilities for the development of tissue models that are functionally similar to our own. 3D bioprinting enables us to create tissues with very specific characteristis – we can match the artificial tissues with real ones in size, organization, cell-cell interactions, and other components of their physiology. This enables us to study tissues like the BBB in great detail, and learn more about specific barrier properties.
By creating effective GBM and BBB artificial models, we would have promising alternatives for testing of therapies for humans, rather than using animals to study some of the crucial concepts. Using these models enables us to study conditions with well defined artificial structures and unique compositions, using less money to manufacture materials and execute the research/trial, and using models specific to patients, leading to more reliable preclinical data.
ADDITIONAL RESOURCES
REFERENCES
Cheng, Hong, et al. “Electrical Stimulation Promotes Stem Cell Neural Differentiation in Tissue Engineering.” Stem Cells International, vol. 2021, 20 Apr. 2021, pp. 1–14, downloads.hindawi.com/journals/sci/2021/6697574.pdf, 10.1155/2021/6697574. Accessed 28 June 2021.
Y. I. Wang, H. E. Abaci, M. L. Shuler, Biotechnol. Bioeng. 2017, 114, 184.
A. Marino, O. Tricinci, M. Battaglini, C. Filippeschi, V. Mattoli, E. Sinibaldi, G. Ciofani, Small 2018, 14, 1702959.Q. T. Ostrom, G. Cioffi, H. Gittleman, N. Patil, K. Waite, C. Kruchko, J. S. Barnholtz-Sloan, Neuro-Oncology 2019, 21, v1; https://do.org/10.1093/neuonc/noz150.Crossref PubMed Web of Science®Google ScholarUniversity of Manchester Library
2M. Koshy, J. L. Villano, T. A. Dolecek, A. Howard, U. Mahmood, S. J. Chmura, R. R. Weichselbaum, B. J. McCarthy, J. Neurooncol. 2012, 107, 207.Crossref PubMed Web of Science®Google ScholarUniversity of Manchester Library
3J. N. Sarkaria, L. S. Hu, I. F. Parney, D. H. Pafundi, D. H. Brinkmann, N. N. Laack, C. Giannini, T. C. Burns, S. H. Kizilbash, J. K. Laramy, K. R. Swanson, T. J. Kaufmann, P. D. Brown, N. Y. R. Agar, E. Galanis, J. C. Buckner, W. F. Elmquist, Neuro-Oncology 2018, 20, 184.
Featured Image Credit Lightspring/Shutterstock
Latest From Instagram
More from the NeuroBlog
How can Neurofilament light concentrations help in Diagnosis?
Overview In May 2022, I attended a conference in Syndey, Australia. The conference was targeted mainly for psychiatrists and neuropsychiatrists. I was able to attend this lovely conference through my company called Monarch Mental Health Group. On the first day, the conference kicked off by an invited keynote speaker. This speaker was a psychiatrist, and…
Execessive Neural Noise in Developmental Dyslexia?
Overview Developmental dyslexia (reading disabilities/disorders, or decoding-based reading disorder) is a neurodevelopmental disorder with multiple potential underlying genetic, neural, and cognitive factors. Past models have not been very successful at integrating key neural and behavioural features of dyslexia with common neural processes, until Hancock et al. (2017) proposed their dyslexia model. Dyslexia risk genes indicate…
Debunking Popular Neuromyths: Do You Use Your Entire Brain?
It is the summer of 2014 on Earth’s Northern hemisphere and the movie Lucy is hitting theatres. The official promotional posters and movie trailers contain the tagline “The average person uses 10% of their brain capacity. Imagine what she could do with 100%.” Lucy becomes the second most successful debut for a French action film,…



