Overview
- Developmental dyslexia (reading disabilities/disorders, or decoding-based reading disorder) is a neurodevelopmental disorder with multiple potential underlying genetic, neural, and cognitive factors.
- Past models have not been very successful at integrating key neural and behavioural features of dyslexia with common neural processes, until Hancock et al. (2017) proposed their dyslexia model.
- Dyslexia risk genes indicate two pathways that result in high neural noise. The first pathway is enhanced/increased glutamatergic signaling (glutamate is the principal excitatory neurotransmitter, released by pyramidal cells), and the second pathway is disrupted neural migration.
- The increased glutamatergic signaling (due to modulations by dyslexia risk genes) disrupts the excitation/inhibition balance of cortical neurons, which eventually leads to increased neural noise.
- The synaptic structure of a cortical microcircuit is important as it can determine its noise characteristics. Any disruptions to the development of typical interconnections can lead to excess neural excitability (hyperexcitability) and result in increased neural noise in the brain.
- In summary, this increased neural noise in dyslexia is linked with the hyperexcitable cortex which may be caused by increased glutamatergic signaling and/or disrupted neural migration.
Developmental dyslexia (reading disabilities/disorders, or decoding-based reading disorder) is neurodevelopmental disorder with multiple potential underlying genetic, neural, and cognitive factors (Pennington, 2006). Castles et al. (2006) had concluded from the analysis of identical and non-identical twins that dyslexia is largely a genetically predisposed reading disorder. Additionally, there is evidence that dyslexia may be more common in boys and this is the case when dyslexia is more severe (Andrewes, 2015). This condition may also have other coexisting specific disabilities such as short-term and working memory difficulties, and also dysgraphia (writing disorder) (Andrewes, 2015). Whilst there are many models that attempt to explain this complex disorder, no model has been able integrate current research which can link key neural and behavioral deficits associated with dyslexia to basic neural processes that is until Hancock et al. (2017) proposed their model for dyslexia.
Hancock et al. combined emerging lines of research and proposed that high neural noise within cortical regions implicated in reading may be a distal contributor to developmental dyslexia. In this article, I will highlight the key points covered in Hancock and colleagues opinion review paper “Neural Noise Hypothesis of Developmental Dyslexia” which was published in 2017.
How does excessive neural noise occur in dyslexia?
According to the authors, dyslexia risk genes indicate two pathways that result in high neural noise. The first pathway is enhanced/increased glutamatergic signaling (glutamate is the principal excitatory neurotransmitter, released by pyramidal cells), and the second pathway is disrupted neural migration. Both of these pathways lead to increased neural noise due to neural hyperexcitability (this results in an imbalance between excitation/inhibition (E/I) in the brain). Hyperexcitability is simply increased excitation in the brain. This idea is similar to what I had covered in my other article “An Imbalance in the Autism Spectrum Disorders Brain” where I had discussed the role of E/I imbalance in autism spectrum disorders.
Isaacson and Scanziani (2011) suggested that balanced levels of E/I within cortical pyramidal-interneuron networks are crucial for the developmental tuning of cortex to sensory input, maintaining neural timing, and also information processing. Therefore, an imbalance in ratio of E/I (hyperexcitability in this case), can disrupt the precise timing of neural activity (Hancock et al., 2017). Note that there are many other reasons why increased neural noise may be present in the brain but the authors focused on the hyperexcitability as the plausible primary source of neural noise in dyslexia. If you are interested more about neural noise in the brain, I recommend that you read this article “Neural variability: friend or foe?” by Dinstein et al. (2015) (spoiler alert: it can be both, depending on the situation!).
The neural noise hypothesis
Hancock et al. (2017) go on to provide evidence for how specific genes in dyslexia result in hyperexcitability due to increased glutamatergic signaling and disrupted neural migration. I have just summarized the main conclusions from this section to make this section short and to the point.
Increased glutamatergic signaling: The main take way here though is that the increased glutamatergic signaling (due to modulations by dyslexia risk genes) disrupts the E/I balance of cortical neurons, subsequently decreasing the precision of spike timing (leading to a hyperexcitable cortex) and increasing neural noise. This increased neural noise is then associated with deficits in rapid auditory processing and reading.
Disrupted neural migration: Both excitatory and inhibitory synaptic connections are required to maintain the E/I balance in local cortical circuits (local cortical circuits contain excitatory pyramidal neurons and inhibitory interneurons). Therefore, the authors report that the synaptic structure of a cortical microcircuit is important as it can determine its noise characteristics and therefore, any disruptions to the development of typical interconnections can lead to excess neural excitability (hyperexcitability) and result in increased neural noise in the brain. While we do not completely understand how disrupted neural migration associated with dyslexia risk genes affect the organization of cortical microcircuits, the authors claim that it is unlikely that the E/I balance is maintained in abnormally migrating circuits. Below is a figure that summarizes the proposals made by Hancock et al. (2017).
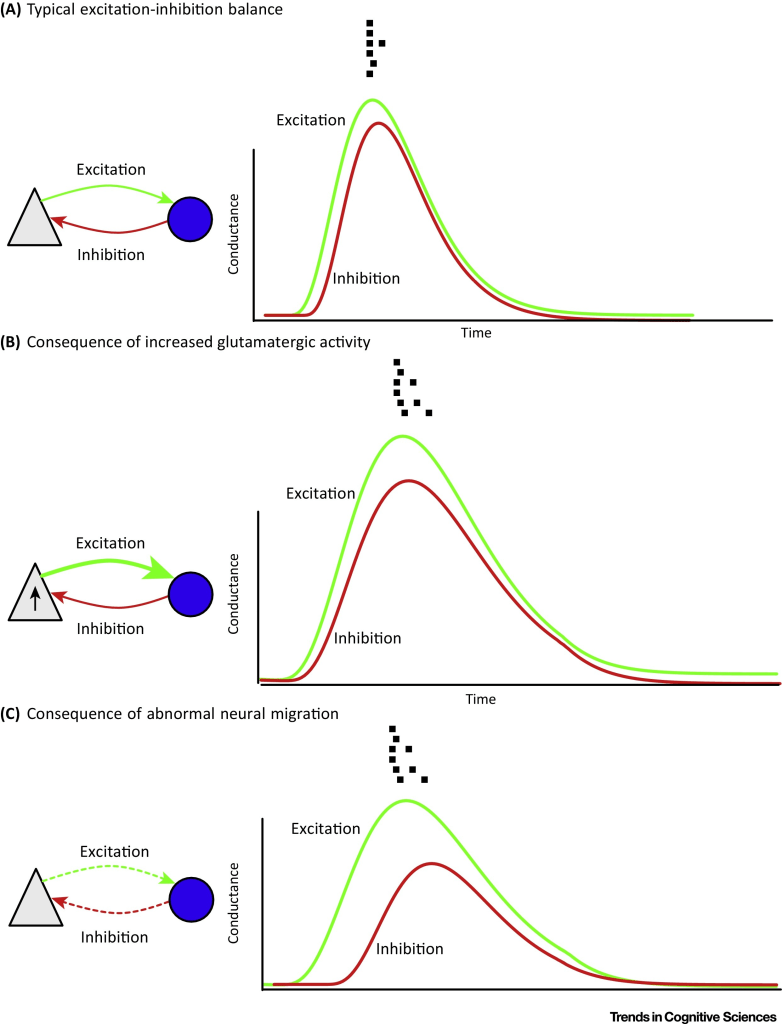
From the figure, image (a) shows typical E/I balance. Note that gray triangles denote excitatory pyramidal cells and blue circles denote inhibitory interneurons (GABAergic). In typical E/I balance, a stimulus that evokes excitatory synaptic conductances (green arrow) is followed by inhibitory conductances (red curves) within few milliseconds. The spike activity (little squares above the graphs) produces neural activity that is precisely timed with respect to the input.
Image (b) shows the consequence of increased glutamatergic signaling due to excitatory post-synaptic activity modulated by dyslexia risk genes (thick green arrow). This results in greater and more temporally extended increase in excitatory conductances and reduced spike timing precision as there is no compensatory increase in feedback inhibition to restore the E/I balance.
Lastly, image (c) shows the consequences of abnormal/disrupted neural migration. Dyslexia risk genes have been linked to disrupted neural migrations and abnormal spike formation of pyramidal cells. This abnormal neural migration can lead to disrupted feedback connectivity (dashed arrows) between inhibitory GABAergic neurons and excitatory pyramidal cells. This leads to reduced feedback inhibition which is required to dampen neural activity and thus, resulting again in temporally extended responses.
In summary, increased neural noise in dyslexia is linked with the hyperexcitable cortex which may be caused due increased glutamatergic signaling and/or disrupted neural migration.
Citations
Andrewes, D. (2015). Neuropsychology: From theory to practice. Psychology Press.
Castles, A., Bates, T., Coltheart, M., Luciano, M., & Martin, N. G. (2006). Cognitive modelling and the behaviour genetics of reading. Journal of Research in Reading, 29(1), 92-103.
Dinstein, I., Heeger, D. J., & Behrmann, M. (2015). Neural variability: friend or foe? Trends in cognitive sciences, 19(6), 322-328.
Hancock, R., Pugh, K. R., & Hoeft, F. (2017). Neural noise hypothesis of developmental dyslexia. Trends in cognitive sciences, 21(6), 434-448.
Isaacson, J. S., & Scanziani, M. (2011). How inhibition shapes cortical activity. Neuron, 72(2), 231-243.
Pennington, B. F. (2006). From single to multiple deficit models of developmental disorders. Cognition, 101(2), 385-413.
Latest From Instagram
More from the NeuroBlog
How can Neurofilament light concentrations help in Diagnosis?
Overview In May 2022, I attended a conference in Syndey, Australia. The conference was targeted mainly for psychiatrists and neuropsychiatrists. I was able to attend this lovely conference through my company called Monarch Mental Health Group. On the first day, the conference kicked off by an invited keynote speaker. This speaker was a psychiatrist, and…
Neurology and Mental Health: The Impact of COVID-19
Overview Coronavirus disease (COVID-19) is an infectious disease caused by the SARS-CoV-2 virus. Most people infected with the virus will experience mild to moderate respiratory illness and recover without requiring special treatment, though some will become seriously ill and require medical attention. Along with growing evidence of neurological symptoms, mental health during the pandemic is…
Inattentional Blindness: An Issue That Even Experts Face!
Overview To make this article more fun, let us first try to perform an interesting task. Below is a random image of a lung CT scan. For the first task, try to count the number of nodules you see in the image below (nodules are the white patches in the black space you see in…



