OVERVIEW
- Gut microbiome are the various microorganisms, such as bacteria and fungi, that live in your digestive tract and is the main location of the human microbiota.
- The gut and brain are connected by the Vagus Nerve, a major component of the autonomic nervous system which enables you to breathe, digest food, and swallow automatically. This nerve is able to send messages to your brain for your colon, and vice versa.
- To support health and digestion, gut microbiota need to be diverse which keeps them balanced. An imbalance in gut bacteria, causes inflammation in the body, which can in turn cause depression, anxiety, mood disorders and changes in the circadian rhythm.
- In more recent studies, it has also been shown that gut microbiome may affect autism, schizophrenia and Parkinson’s disease.
The Microbiome-Gut-Brain Axis
Trillions of bacteria exist in the human intestine and have been shown to play a vital role in gut-brain communication through their influence on nerve, immune and endocrine pathways. It has been shown that there are significant differences in the composition of the gut microbiome in patients with various mental disorders, including depression, bipolar disorder, schizophrenia, and Autism Spectrum Disorder (ASD). Improving the beneficial bacteria in the intestines, such as through the use of probiotics, prebiotics, or dietary changes, may improve the mood of healthy people and patient groups and reduce anxiety.
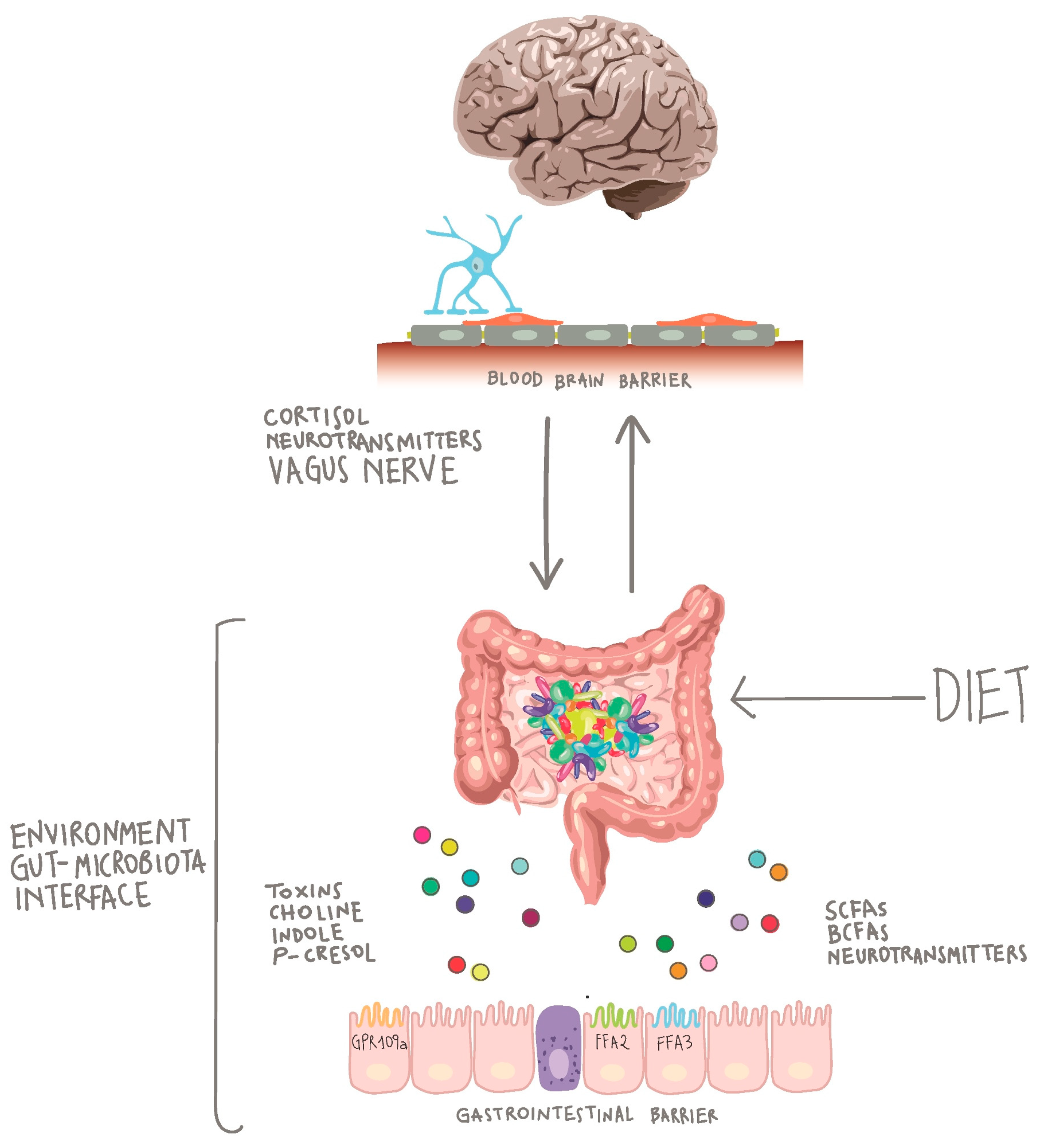
Gastrointestinal symptoms often appear in mental health disorders. Loss of appetite and weight changes are common features of major depressive disorder (MDD), while diarrhea and nausea are common complaints in people experiencing anxiety disorders. Gastrointestinal problems often coexist with autism spectrum disorder (ASD), schizophrenia, and Parkinson’s disease. The mechanism by which our intestinal bacteria communicate and influence the central nervous system is gradually being discovered, and it spans the nervous, endocrine and immune systems. There is a surprising overlap between the pathways affected by the microbiome and the pathways related to mental illness.
The gut microbiome has been shown to play an important role in the development and function of the hypothalamus-pituitary-adrenal (HPA) axis. The HPA axis mediates stress responses and plays an important role in various mental illnesses, especially depression and anxiety. Our intestinal bacteria also significantly affect the immune system and may be related to immune dysfunction, which is characteristic of some disorders such as depression and schizophrenia. Interestingly, the gut microbiome also affects neurotransmission. In addition to the direct production of various neurotransmitters, norepinephrine, dopamine and gamma-aminobutyric acid, intestinal bacteria have been shown to regulate tryptophan metabolism and serotonin production.
Table 1 further explores the communication pathways between the microbiome, the gut and the brain, and their relevance to mental illness
| Gut-Brain-Axis Pathway of Communication | Link with Psychiatric Disorders |
| Immunomodulation: Gut microbiome plays an important role in immune system development. The translocation of microorganisms from the intestine to the blood is usually prevented by the very adherent internal epithelium. However, stress has been shown to change the integrity of the barrier, and the “leaky gut” may allow microorganisms to move out of the intestines, causing inflammation. | Many mental illnesses are associated with low-grade chronic inflammation and elevated pro-inflammatory cytokines. The source of this is unknown. Gut microbiota disorders may represent a possible mechanism, combining chronic stress, “gut leakage,” and cytokines. Neuropsychiatric and producer disorders are associated with depression. |
| Stress response: Hypothalamus pituitary adrenal (HPA) axis mediates the stress response through cascade interactions in the culminang cortisol product. There is substantial evidence that the gut microbiome is a key regulator of this stress pathway. Various probiotics and prebiotics have been proven to reduce cortisol, and therefore stress, in healthy people. | Adverse events and chronic stress in early life are major risk factors for depression, anxiety and other mental illnesses |
| Production of nerve infiltrating substances: Intestinal bacteria can directly produce neurotransmitters used in the human body, including GABA, serotonin, norepinephrine, acetylcholine and dopamine. In addition, they produce short-chain acids (SCFA), such as butyrate, which is believed to be important to brain health. | GABA, serotonin, norepinephrine, acetylcholine and dopamine are of course very implicated in various mental illnesses. However, the quantities produced by bacteria are relatively small, and is unlikely to directly affect human neurotransmission to a large extent. Butyrate has shown anti-mania and depression effects in animal models. In preclinical studies of Huntington’s, Parkinson’s, and Alzheimer’s disease, it has also been shown to be beneficial. |
| Tryptophan and serotonin metabolism: Tryptophan is an essential amino acid. Although it is best known for its role as a precursor to serotonin, most of it is actually metabolized via the alternative pathway (kynurenine pathway). This pathway leads to the products of neuroactive compounds, such as kynurenine, kynurenic acid (KA), and quinolinic acid (QA). The gut microbiota appears to control the host’s tryptophan metabolism along this kynurenine pathway, thereby increasing the production of neuroactive KA and QA, while reducing the amount of tryptophan available for serotonin synthesis. | Serotonin may be the most studied of all the neurotransmitters. It is involved in mental illnesses, especially mood and anxiety disorders. However, the kynurenine pathway may be equally important in the pathogenesis of depression. Kynurenine and QA appear to have inhibitory effects, while KA has neuroprotective properties. The imbalance of these metabolites may be related to pathogenesis of depression. |
Current Evidence of the Role of the Microbiome
BIPOLAR AFFECTIVE DISORDER (BPAD)
Several studies have investigated the microbiome composition of people with BPAD. The first was a relatively large study involving 115 participants and reported a decrease in the level of faecalis. This finding was repeated in an Austrian study of 32 participants with bipolar disorder and proved to be consistent with an MDD study in which similar bacteria were reported to be underrepresented. However, a Danish study compared the diagnosis of unaffected first-degree relatives and healthy individuals in the microbiome of 113 participants with a recent BPAD diagnosis and found no difference in faecalis. They report that Flavonifractor is a genus of bacteria that can induce oxidative stress and inflammation, and is associated with bipolar disorder.
MAJOR DEPRESSIVE DISORDER (MDD)
There are common features in the microbiome of depressed individuals as compared to healthy controls, however evidence is not conclusive as there is significant variability between individuals. One large-scale population study which used data from the Flemish Gut Flora Project to investigate the relationship between the microbiome, quality of life, and depression, found that two bacterial groups, namely Coprococcus and Dialister, were depleted in participants with an MDD diagnosis regardless of their antidepressant treatment. The pathophysiology of depression involves neuroimmune-neuroendocrine dysregulation. However, the extent to which the gut microbiota composition and function mediate the dysregulation of these pathways is largely unknown. The role of the microbiome in MDD is supported by an interesting finding, whereby microbiome colonies of depressed individuals are introduced in healthy mice, subsequently the mice start showing depressive-like symptoms.
NEURODEGENERATIVE DISEASES
Although Parkinson’s disease (PD) is the most studied disease, the microbiome is implicated in a variety of neuronal diseases, including Alzheimer’s disease (AD), multiple sclerosis, and amyotrophic lateral sclerosis. Although animal and translational models have incriminated a disturbed gut microbiota in a number of CNS disorders, including Parkinson’s disease, evidence from human clinical trials is lacking.
Several studies have found that Lactobacillus, Bifidobacterium, Akkermania, and Verrucomicrobia may be increased in Parkinson’s, while Faecalis, Faecoccus, Brautia and Prevotella may be underrepresented. In contrast, some evidence proposes that bifidobacteria may be decreased in AD. Interestingly, the composition of the microbiome in Parkinson’s is surprisingly similar to that seen in idiopathic rapid eye movement sleep behavior disorder (a disease that is considered a prodromal symptom of PD), which suggests that changes in the microbiome may precede PD in the development of symptoms.
It is important to note that the MGB axis has sparked a lot of interest and research in recent years, however results are largely tentative. There are significant differences between the unique internal environments of individuals, and a lack of large scale clinical trials that would enable us to draw conclusive evidence.
CONCLUSION
The MGB axis provides a new, much-needed paradigm for psychiatry to address mental illness. Even by using a holistic biopsychosocial approach to treatment of mental health diagnoses, many individuals experience chronic psychological distress. Although much remains to be discovered about the interaction of the gut microbiome and health, particularly its effects on brain and mental health, the fields of nutrition and gut health are beginning to become an important part of overall psychiatric care. As developed countries become more aware of healthy dietary intake, integrating dietary changes, and other microbiome-based interventions, into psychological and psychiatric practise, may be a plausible step forward.
VIDEO RESOURCES
Latest From Instagram
REFERENCES
Belkaid, Yasmine, and Timothy W. Hand. “Role of the Microbiota in Immunity and Inflammation.” Cell, vol. 157, no. 1, Mar. 2014, pp. 121–141, pubmed.ncbi.nlm.nih.gov/24679531/, 10.1016/j.cell.2014.03.011. Accessed 25 Aug. 2021.
Bourassa, Megan W., et al. “Butyrate, Neuroepigenetics and the Gut Microbiome: Can a High Fiber Diet Improve Brain Health?” Neuroscience Letters, vol. 625, June 2016, pp. 56–63, pubmed.ncbi.nlm.nih.gov/26868600/, 10.1016/j.neulet.2016.02.009. Accessed 25 Aug. 2021.
Butler, Mary I., et al. “The Gut Microbiome and Mental Health: What Should We Tell Our Patients?: Le Microbiote Intestinal et La Santé Mentale : Que Devrions-Nous Dire à Nos Patients?” The Canadian Journal of Psychiatry, vol. 64, no. 11, 17 Sept. 2019, pp. 747–760, http://www.ncbi.nlm.nih.gov/pmc/articles/PMC6882070/, 10.1177/0706743719874168. Accessed 25 Aug. 2021.
Dickerson, Faith, et al. “The Microbiome, Immunity, and Schizophrenia and Bipolar Disorder.” Brain, Behavior, and Immunity, vol. 62, May 2017, pp. 46–52, pubmed.ncbi.nlm.nih.gov/28003152/, 10.1016/j.bbi.2016.12.010. Accessed 25 Aug. 2021.
Dinan, T G, et al. “Genomics of Schizophrenia: Time to Consider the Gut Microbiome?” Molecular Psychiatry, vol. 19, no. 12, 7 Oct. 2014, pp. 1252–1257, pubmed.ncbi.nlm.nih.gov/25288135/, 10.1038/mp.2014.93. Accessed 25 Aug. 2021.
Foster, Jane A., et al. “Stress & the Gut-Brain Axis: Regulation by the Microbiome.” Neurobiology of Stress, vol. 7, Dec. 2017, pp. 124–136, pubmed.ncbi.nlm.nih.gov/29276734/, 10.1016/j.ynstr.2017.03.001. Accessed 25 Aug. 2021.
O’Mahony, S.M., et al. “Serotonin, Tryptophan Metabolism and the Brain-Gut-Microbiome Axis.” Behavioural Brain Research, vol. 277, Jan. 2015, pp. 32–48, pubmed.ncbi.nlm.nih.gov/25078296/, 10.1016/j.bbr.2014.07.027. Accessed 25 Aug. 2021.
Privitera, Gregory J., et al. “From Weight Loss to Weight Gain: Appetite Changes in Major Depressive Disorder as a Mirror into Brain-Environment Interactions.” Frontiers in Psychology, vol. 4, 2013, pubmed.ncbi.nlm.nih.gov/24312070/, 10.3389/fpsyg.2013.00873. Accessed 25 Aug. 2021.
Quigley, Eamonn M. M. “Microbiota-Brain-Gut Axis and Neurodegenerative Diseases.” Current Neurology and Neuroscience Reports, vol. 17, no. 12, 17 Oct. 2017, pubmed.ncbi.nlm.nih.gov/29039142/, 10.1007/s11910-017-0802-6. Accessed 25 Aug. 2021.
Rea, Kieran, et al. “The Microbiome: A Key Regulator of Stress and Neuroinflammation.” Neurobiology of Stress, vol. 4, Oct. 2016, pp. 23–33, pubmed.ncbi.nlm.nih.gov/27981187/, 10.1016/j.ynstr.2016.03.001. Accessed 25 Aug. 2021.
Resende, Wilson R., et al. “Effects of Sodium Butyrate in Animal Models of Mania and Depression.” Behavioural Pharmacology, vol. 24, no. 7, Oct. 2013, pp. 569–579, pubmed.ncbi.nlm.nih.gov/23994816/, 10.1097/fbp.0b013e32836546fc. Accessed 25 Aug. 2021.
Shen, Yang, et al. “Analysis of Gut Microbiota Diversity and Auxiliary Diagnosis as a Biomarker in Patients with Schizophrenia: A Cross-Sectional Study.” Schizophrenia Research, vol. 197, July 2018, pp. 470–477, pubmed.ncbi.nlm.nih.gov/29352709/, 10.1016/j.schres.2018.01.002. Accessed 25 Aug. 2021.
Verduci, Elvira, et al. “Nutrition, Microbiota and Role of Gut-Brain Axis in Subjects with Phenylketonuria (PKU): A Review.” Nutrients, vol. 12, no. 11, 29 Oct. 2020, p. 3319, http://www.mdpi.com/2072-6643/12/11/3319/htm, 10.3390/nu12113319. Accessed 23 Aug. 2021.
Wallace, Caroline J. K., and Roumen Milev. “The Effects of Probiotics on Depressive Symptoms in Humans: A Systematic Review.” Annals of General Psychiatry, vol. 16, no. 1, 20 Feb. 2017, pubmed.ncbi.nlm.nih.gov/28239408/, 10.1186/s12991-017-0138-2. Accessed 25 Aug. 2021.
Featured Image Credit Kateryna Kon / Shutterstock
More from the NeuroBlog
How can Neurofilament light concentrations help in Diagnosis?
Overview In May 2022, I attended a conference in Syndey, Australia. The conference was targeted mainly for psychiatrists and neuropsychiatrists. I was able to attend this lovely conference through my company called Monarch Mental Health Group. On the first day, the conference kicked off by an invited keynote speaker. This speaker was a psychiatrist, and…
Execessive Neural Noise in Developmental Dyslexia?
Overview Developmental dyslexia (reading disabilities/disorders, or decoding-based reading disorder) is a neurodevelopmental disorder with multiple potential underlying genetic, neural, and cognitive factors. Past models have not been very successful at integrating key neural and behavioural features of dyslexia with common neural processes, until Hancock et al. (2017) proposed their dyslexia model. Dyslexia risk genes indicate…
Debunking Popular Neuromyths: Do You Use Your Entire Brain?
It is the summer of 2014 on Earth’s Northern hemisphere and the movie Lucy is hitting theatres. The official promotional posters and movie trailers contain the tagline “The average person uses 10% of their brain capacity. Imagine what she could do with 100%.” Lucy becomes the second most successful debut for a French action film,…



