OVERVIEW
- The blood brain barrier is the complex network of brain capillaries and surrounding glia that governs the homeostatic flow of ions, metabolites, and molecules between the brain and the blood.
- A pathological or damaged blood brain barrier has high permeability and low integrity, which results in a greater flow of solutes and metabolites that can enter the neural tissue – but it also results with solutes and metabolites exiting the brain into the blood. Specific solutes and metabolites are biomarkers of blood brain barrier integrity.
- Examples of biomarkers include everything from pulse and blood pressure, to the amount of specific proteins analysed in urine. They are objective, quantifiable characteristics of biological processes, which is extremely important in health research. See examples of biomarkers of a COVID19 infection in the article below.
- Pathological conditions affect the Blood Brain Barrier and associated biomarkers.
- For example, the astrocytic protein S100B has been examined as a peripheral biomarker of BBB permeability and gliosis, associated with Traumatic Brain Injury.
- Amyloid beta (Aβ) is one of the main constituents of the amyloid plaques found in the brain of people with Alzheimer’s disease.
- Also read about the role of miRNA as biomarkers for various conditions including stroke and epilepsy!
Recap on the Blood Brain Barrier
So, as you may have noticed, this post will be discussing the biomarkers of blood brain barrier integrity! I will be touching on some concepts that have been described in my previous post – so if you haven’t read it, quickly check it out before reading this. Here is a quick recap: The adult central nervous system is comprised of three anatomical barriers: the blood-cerebrospinal fluid barrier, the blood brain barrier, and the arachnoid barrier (Lindblad et al., 2020). These barriers help protect important neural tissue from damage and maintain homeostasis of ion flux into and out of the brain. Today, I will be discussing in greater detail the blood brain barrier, and the biomarkers of changes in blood brain barrier integrity that are associated with different pathologies.
The blood brain barrier is the complex network of brain capillaries and surrounding glia that governs the homeostatic flow of ions, metabolites, and molecules between the brain and the blood (Janigro et al., 2021). The capillaries are comprised of highly-selective, semipermeable linings of endothelial cells which act as the main ‘barrier’ component of the blood brain barrier. Pericytes, astrocytic endfeet, and basement membranes wrap around the endothelial cells, securing them in place and assisting in the prevention of ion and metabolite flux from the blood to the brain. This prevention of solute flux corresponds to a low permeability of the blood brain barrier.
As mentioned above, a heathy blood brain barrier has a low permeability to solute flux. I use the term permeability synonymously with integrity – they are both used to describe how readily solutes diffuse across its membrane. High integrity represents the capacity of the barrier to limit the flow of molecules, whereas low integrity represents a limited capacity, resulting in a larger flux of molecules into the neural tissue. The opposite is true for permeability – high permeability results in a large flux of molecules into the neural tissue, whereas low permeability represents a low flux of molecules into the neural tissue.
Simply, an effective, healthy blood brain barrier has low permeability and high integrity. A pathological or damaged blood brain barrier has high permeability and low integrity, which results in a greater flow of solutes and metabolites that can enter the neural tissue – but it also results with solutes and metabolites exiting the brain into the blood. Specific solutes and metabolites are biomarkers of blood brain barrier integrity.
What is a Biomarker
The term ‘biomarker’ is defined as “any substance, structure, or process that can be measured in the body or its products and influence or predict the incidence of outcome or disease” (World Health Organisation, 2001). Biomarkers are often used in health risk assessments as prognostic or diagnostic indicators of disease manifestation, progression, or both, as well as response to therapy (Ptolemy & Rifai, 2010). Examples of biomarkers include everything from pulse and blood pressure, to the amount of specific proteins analysed in urine. They are objective, quantifiable characteristics of biological processes, which is extremely important in health research.
For example, some biomarkers of COVID-19 infection include a:
- Decreased lymphocyte count
- Decreased platelet count
- Increased blood urea nitrogen
- Decreased albumin
- Increased creatinine (Ponti et al., 2020)
Researchers and clinicians can measure these aforementioned biomarkers and be fairly certain of their COVID-19 diagnoses. This is similar with biomarkers of blood brain barrier integrity – researchers are able to quantify specific proteins that exit the neural tissue into the peripheral circulation and make diagnoses on a wide array of pathological conditions that effect the blood brain barrier and brain. Different types of metabolites will be present in the peripheral circulation according to pathological condition that has damaged the blood brain barrier. The next section will describe what diseases effect the integrity of the blood brain barrier and what biomarkers can be measured to help us diagnose the disease.
Pathological Conditions That Affect The Blood Brain Barrier and Their Biomarkers
Peripheral biological fluids (urine, blood, plasma, serum, saliva) represent suitable matrices to detect and quantify brain-derived proteins that correspond to changes in BBB permeability and glio-neuronal damage (Janigro et al., 2021). According to Janigro et al. (2021), peripheral biomarker proteins must:
(i) be present in brain interstitial fluids or be released by neurovascular cells into the interstitial or perivascular spaces, reaching the peripheral blood across a leaky BBB or by cerebrospinal fluid–blood exchange;
(ii) have a concentration gradient driving passive diffusion;
(iii) have a known and appropriate half-life to allow diagnostic interpretation; and
(iv) have a low molecular weight to allow a rapid egress across the damaged barriers or interfaces.
In the subsequent sections, I will outline different brain pathologies and their corresponding protein biomarkers. Because of similarities in the degradative processes of some of these pathologies, you may notice that some biomarkers may appear more than once.
Traumatic Brain Injury

Much of the neurological clinical biomarker literature is focused on traumatic brain injury (TBI). TBI is a disruption in the normal function of the brain, including a massive spike in blood brain barrier permeability, that is caused by a bump, blow, or jolt to the head. TBI occurs often in sport and recreational activities. The astrocytic protein S100B has been examined as a peripheral biomarker of BBB permeability and gliosis, associated with TBI. Early studies showed blood S100B levels to rapidly increase in response to a sudden increase in BBB permeability, and therefore supported its clinical use for TBI (Janigro et al., 2021). S100B has been reported reported to rule out TBI sequelae in emergency room settings with the measurement of blood S100B levels displaying a 99.7% negative predictive value (Janigro et al., 2021). In sports, S100B blood levels increased immediately after American football games as compared to pregame baselines in players experiencing repeated head hits (Janigro et al., 2021).
Alzheimer’s Disease
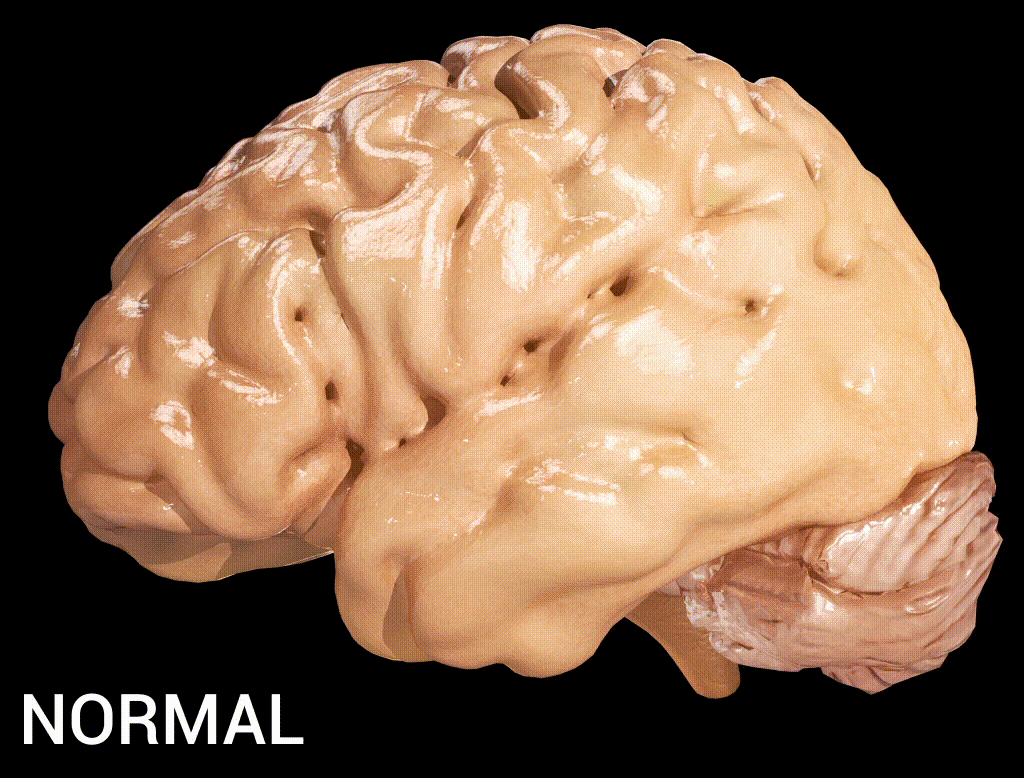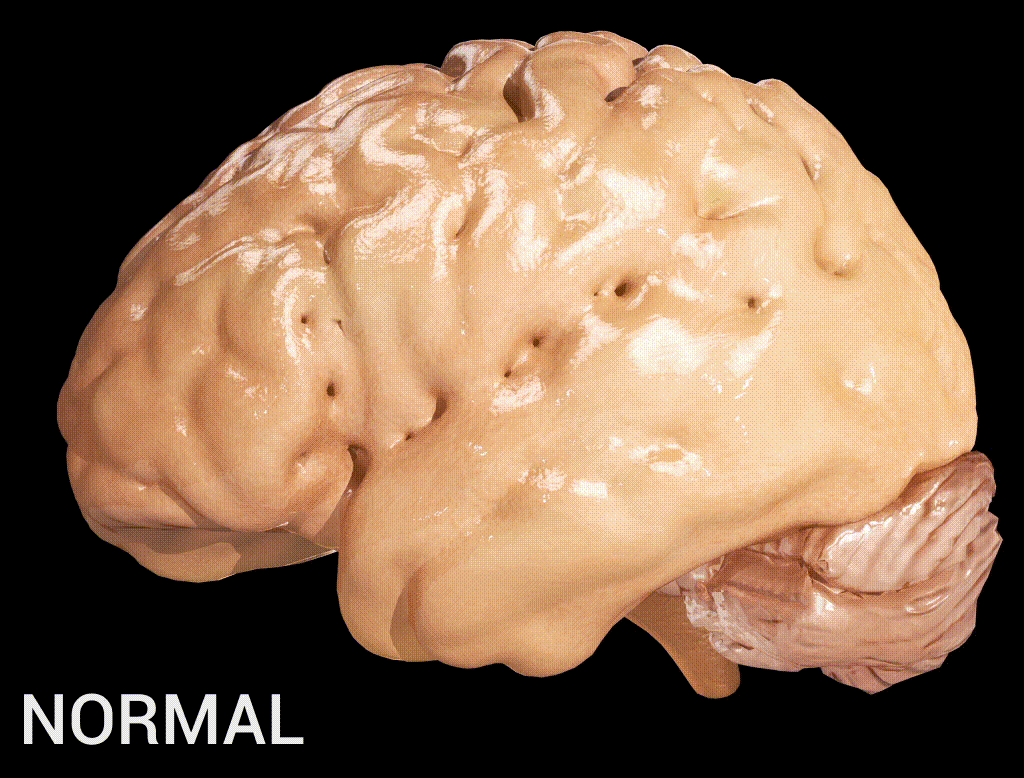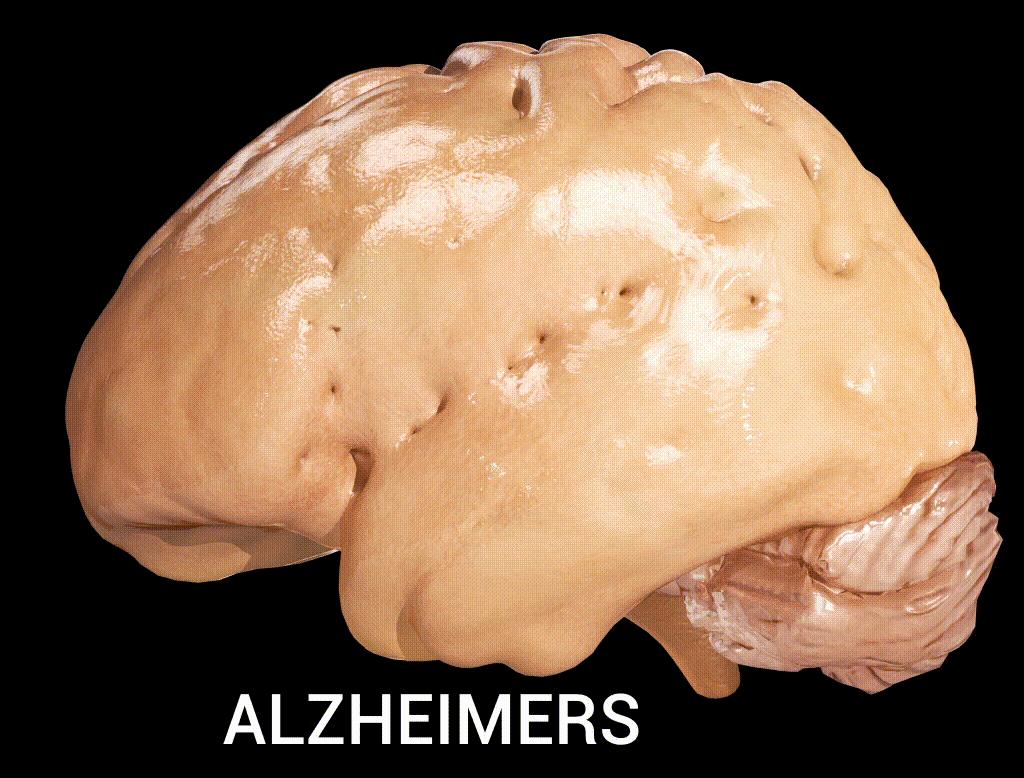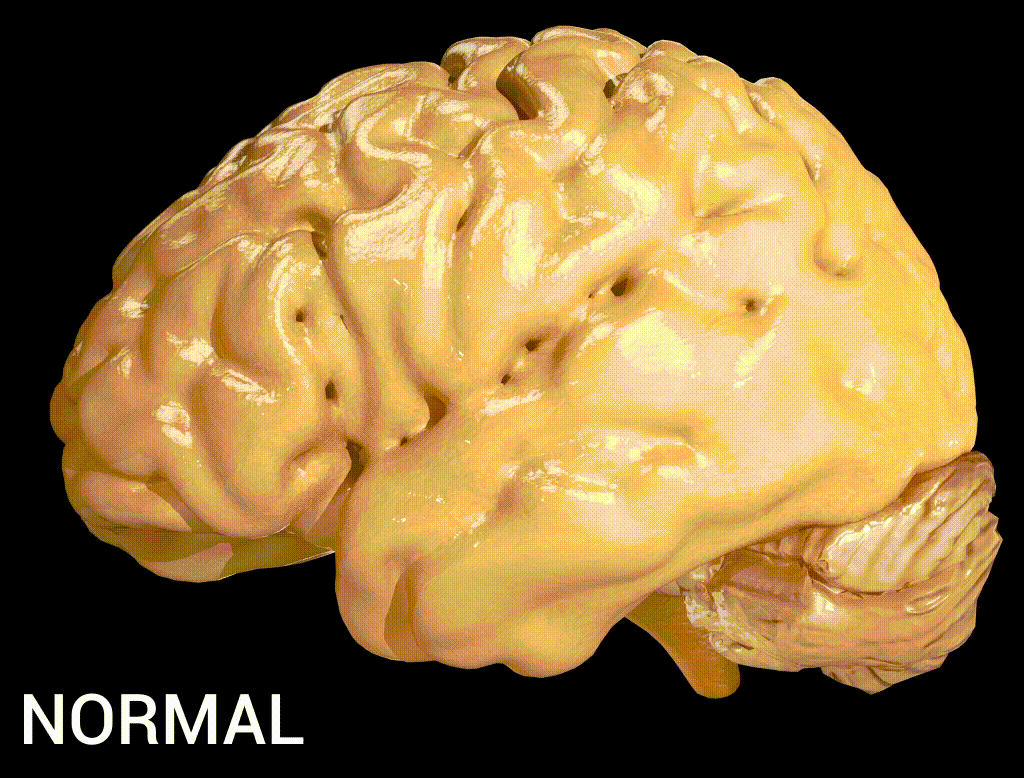
Alzheimer’s disease (AD) is a neurogenerative disorder that deteriorates neurons, resulting in a loss of memory and cognition. Amyloid beta (Aβ), a 36–43 amino acids peptide, is one of the main constituents of the amyloid plaques found in the brain of people with Alzheimer’s disease (Kadry et al., 2020). The high levels of accumulated β-amyloid protein in the brain of people with Alzheimer’s activate microglia and astrocytes, which lead to a cascade of events producing toxic molecules, neuronal damage, and synaptic dysfunction (Kadry et al., 2020).
Accumulating evidence points to blood phosphorylated tau as a promising biomarker to improve the diagnosis and staging of Alzheimer’s disease (Janigro et al., 2021). In a cross-sectional study performed in AD patients, phosphorylated tau isoforms were used as diagnostic biomarkers to track disease progression (Janigro et al., 2021). A method measuring concentrations of tau isoforms in plasma was implemented using stable isotope labeling kinetics and mass spectroscopy (Janigro et al., 2021).
Note. From Alzheimer’s Disease, by User:DrJanaOfficial, 2016, Wikimedia (https://commons.wikimedia.org/wiki/File:Alzheimer%27s_Disease.gif). CC BY 4.0
Ischemic Stroke
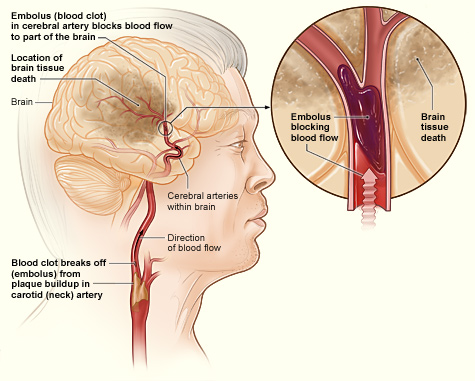
Stroke is a common neurological disease that occurs when the blood supply to the brain is interrupted, resulting in a shortage of oxygen (hypoxia) and nutrients to brain tissue (Vijayan & Reddy, 2016). There are extensive studies showing the effect of hypoxia–ischemia on the BBB, which suggested the disruption of the TJs and the increase in BBB permeability (Kadry et al., 2020). It has been hypothesises that Micro RNAs (MiRNA) may be used as a biomarker for ischemic strokes.
MiRNAs are composed of a group of endogenous and small non-protein coding genes present in virtually all animals, plants and some viruses (Vijayan & Reddy, 2016). According to the miRbase-21 database released in June 2014, 1881 precursor and 2588 mature miRNAs have been identified (www.mirbase.org) in such human diseases as cancer, viral infection, diabetes, immune-related diseases, and neurodegenerative disorders (Vijayan & Reddy, 2016).
Whole blood and plasma are two matrices which can be assessed to detect circulatory MiRNAs changes. Using polymerase chain reaction (PCR) testing, Zeng et al. (2011) found that the level of miR-210 in blood drawn from stroke patients was significantly higher than in blood samples from patients who never had a stroke. Also using PCR, Wang et al. (2014) discovered an upregulation of the MiRNAs miR-106b-5P, miR-4306, miR-320e, and miR-320d in plasma. These findings suggest that measuring circulatory MiRNA in both blood and plasma samples from acute IS patients might be useful in diagnosing and prognosing stroke, and it might also be useful in predicting the response of stroke patients (Vijayan & Reddy, 2016).
Epilepsy
Epilepsy is a disorder of the brain characterised by an enduring predisposition to generate epileptic seizures (Duncan et al., 2006) Epilepsy affects people from all ages, ranging from neonates to elderly people, and has varied causes and manifestations, with many distinct seizure types and several identifiable syndromes (Duncan et al., 2006). It is a CNS pathological condition that significantly effects the BBB.
As highlighted above, MiRNAs have been recognized as a therapeutic target and a diagnostic tool in multiple neurological disorders, because they can be can be detected in the blood after crossing BBB in a form of miRNA containing exosomes (Chmielewska et al., 2018). Serum miRNA concentration has been analyzed in humans with epilepsy. Studies have revealed that miRNA has been differentially expressed in epilepsy patients compared to healthy control, with one of the miRNAs, named hsa-miR106b-5p, being established as a high sensitivity and specificity marker for epilepsy diagnosis (Chmielewska et al., 2018). Additionally, in drug resistant epilepsy, the miRNAs hsa-miR301a-3p has been shown to have the biggest diagnostic value for drug resistant epilepsy (Chmielewska et al., 2018).
Summary
Biomarkers are often in research and medicine as a measure for prognostic or diagnostic indicators. Under pathological conditions, the blood brain barrier undergoes changes in its integrity, resulting in a sudden change in solutes, metabolites, and proteins in the blood. These changes in solutes and proteins are biomarkers that can be used to diagnose specific brain pathologies. Proteins of interest include S100B amyloid-b which have been used to diagnose traumatic brain injury and Alzheimer’s disease, respectively. MicroRNAs have also been used as a diagnostic biomarker for brain pathologies that affect the blood brain barrier including ischemic stroke and epilepsy. And there you have it – a summary on just a fraction of all of the biomarkers that are out there available for the diagnosis of different brain pathologies that are used in both research and medicine.
Additional Resources
Latest From Instagram
References
Chmielewska, N., Szyndler, J., Makowska, K., Wojtyna, D., Maciejak, P., & Płaźnik, A. (2018). Looking for novel, brain-derived, peripheral biomarkers of neurological disorders. Neurologia i neurochirurgia polska, 52(3), 318-325.
Duncan, J. S., Sander, J. W., Sisodiya, S. M., & Walker, M. C. (2006). Adult epilepsy. The Lancet, 367(9516), 1087-1100.
Kadry, H., Noorani, B., & Cucullo, L. (2020). A blood–brain barrier overview on structure, function, impairment, and biomarkers of integrity. Fluids and Barriers of the CNS, 17(1), 1-24.
Janigro, D., Bailey, D. M., Lehmann, S., Badaut, J., O’flynn, R., Hirtz, C., & Marchi, N. (2021). Peripheral Blood and Salivary Biomarkers of Blood–Brain Barrier Permeability and Neuronal Damage: Clinical and Applied Concepts. Frontiers in Neurology, 11, 1767.
Lindblad, C., Nelson, D. W., Zeiler, F. A., Ercole, A., Ghatan, P. H., von Horn, H., … & Thelin, E. P. (2020). Influence of blood–brain barrier integrity on brain protein biomarker clearance in severe traumatic brain injury: a longitudinal prospective study. Journal of neurotrauma, 37(12), 1381-1391.
Vijayan, M., & Reddy, P. H. (2016). Peripheral biomarkers of stroke: focus on circulatory microRNAs. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 1862(10), 1984-1993.
Wang, W., Sun, G., Zhang, L., Shi, L., & Zeng, Y. (2014). Circulating microRNAs as novel potential biomarkers for early diagnosis of acute stroke in humans. Journal of Stroke and Cerebrovascular Diseases, 23(10), 2607-2613.
Zeng, L., Liu, J., Wang, Y., Wang, L., Weng, S., Tang, Y., … & Yang, G. Y. (2011). MicroRNA-210 as a novel blood biomarker in acute cerebral ischemia. Front Biosci (Elite Ed), 3(3), 1265-1272.
Featured Image Credit Naeblys / Shutterstock
More from the NeuroBlog
How can Neurofilament light concentrations help in Diagnosis?
Overview In May 2022, I attended a conference in Syndey, Australia. The conference was targeted mainly for psychiatrists and neuropsychiatrists. I was able to attend this lovely conference through my company called Monarch Mental Health Group. On the first day, the conference kicked off by an invited keynote speaker. This speaker was a psychiatrist, and…
Execessive Neural Noise in Developmental Dyslexia?
Overview Developmental dyslexia (reading disabilities/disorders, or decoding-based reading disorder) is a neurodevelopmental disorder with multiple potential underlying genetic, neural, and cognitive factors. Past models have not been very successful at integrating key neural and behavioural features of dyslexia with common neural processes, until Hancock et al. (2017) proposed their dyslexia model. Dyslexia risk genes indicate…
Debunking Popular Neuromyths: Do You Use Your Entire Brain?
It is the summer of 2014 on Earth’s Northern hemisphere and the movie Lucy is hitting theatres. The official promotional posters and movie trailers contain the tagline “The average person uses 10% of their brain capacity. Imagine what she could do with 100%.” Lucy becomes the second most successful debut for a French action film,…



